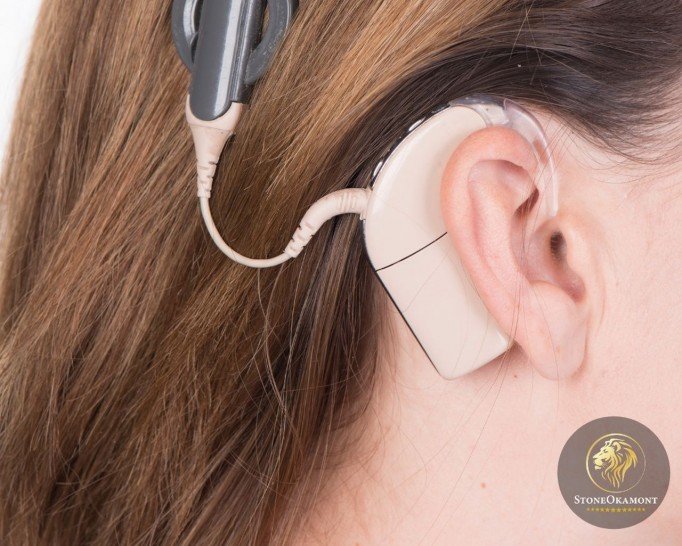
Known as bionic ear, the cochlear implant is an electronic device for people with hearing loss. It’s used to restore hearing function in hearing impaired people.
The device contains two parts, the external sound processor and the surgical implant. The outside of the device consists of a speech processor, a transmitting antenna and a microphone, that’s, the sound is picked up and transmitted to the antenna. The internal part works by means of radio frequency, that’s, it receives the information that’s transmitted by the antenna.
The cochlear implant sends the received signal directly to the auditory nerve. It captures sound, processes and electrically stimulates the nerve, this means that the equipment can partially replace damaged cells of the cochlea, located in the internal ear. Remembering that only the audiologist can determine which candidates for cochlear implant.
ANVISA classifies cochlear implants in the category of correlatos with risk classification III.
Prior to the marketing of this product, it’ll be necessary to obtain the following operating license, operating authorization, certificate of good manufacturing practices and product registration. Stone Okamont specializes in company and product regularization at Anvisa.
Request a budget
Operating License
Before you file a cochlear implant registration application with ANVISA, the first step is to obtain the Operating License for your establishment. It’s acquired from a survey conducted by VISA, the Local Sanitary Surveillance.
The License is a documentation that ascertains whether the establishment has technical and operational conditions to carry out its activity.
Operating Authorization
The AFE, Authorization for the Operation of Companies, is mandatory to register health materials in ANVISA. It’s granted by the National Health Surveillance Agency, upon publication in the Official Gazette, after analysis and approval of the petition sent by the company.
Obtaining AFE means that the company operates in accordance with Brazilian legislation and is able to operate nationwide.
Good Manufacturing Practices
The Certificate of Good Manufacturing Practices (CBPF) is the document that attests to compliance with the guidelines of the legislation in force for the manufacture of a given product. It’s issued to prove that the company has been instructed and inspected during the manufacturing process.
The Certification of Good Practices, besides being a requirement, is one of the most improved parameters in the world, attesting that the establishment complies with good manufacturing practices. The risk classification products III and IV must need the Certificate of Good Practices with ANVISA.
How can we help?
We operate exclusively, adjusting, elaborating and implementing all the necessary procedures for the implementation of the quality system according to the needs of each client.
We have specialized professionals in all areas, allowing you to be appointed an expert with adequate knowledge for your field of activity, so that this process flows in an agile way and avoiding rework.






