Do you know what it takes to regularize a colostomy bag at Anvisa? In this article we will talk about what a colostomy bag is, what its function is and how to notify it. Check out!
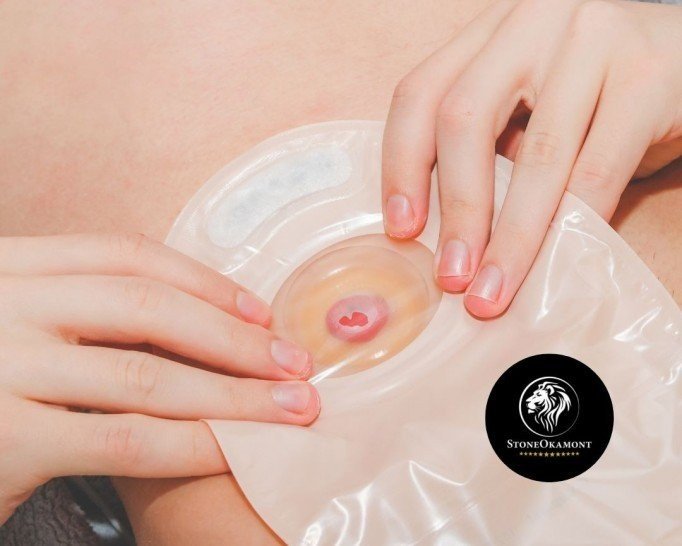
The colostomy, a disease that is little talked about, but is nothing more than the exteriorization of a part of the large intestine through the abdominal wall, to redirect intestinal transit.
The location of the colostomy bag will depend on the portion of intestine affected. The use of the colostomy bag can be temporary, if there is the possibility of reconstructing the intestinal transit.
In order to regularize the colostomy bag, the company must obligatorily have a Sanitary License and Company Operating Permit from Anvisa for health products, as this product is a correlate.
What is needed to regularize the colostomy bag?
Before notifying the colostomy bag or any other product at Anvisa, you must first regularize your company. In a more simplified form, it is necessary to carry out the licensing steps of the establishment to be able to regularize the products, or specifically in this case, to notify the colostomy bag.
The first step is to regularize the company with VISA – Local Sanitary Surveillance. In order to obtain the sanitary license, the company must meet the requirements of Good Manufacturing Practices and/or Distribution and be in compliance with other applicable regulatory bodies, such as an environmental license, if applicable, a Fire Department Inspection Notice (AVCB), business license from the city hall, etc. Sanitary Surveillance inspectors will carry out an on-site inspection to attest to compliance with the sanitary requirements.
The second step is to register the company with Anvisa and provide proof of size.
The third step to regularize the colostomy bag is to obtain the Operating Permit – AFE from ANVISA – National Health Surveillance Agency. At this stage, it is necessary to assemble the dossier and file the AFE request.
What is the risk classification of the colostomy bag?
To regularize the colostomy bag, it is necessary to classify it according to the risk class
The colostomy bag fits risk classification I according to RDC 185/2001, so its regularization is through notification at Anvisa. Although the notification is a simpler process than the registration process, it is necessary to pay attention to the documents required to assemble the notification dossier. Companies of risk classes I and II products do not need the Certificate of Good Manufacturing Practices (CBPF), however it is necessary to comply with the requirements of Good Practices.
The CBPF is required for companies of risk class III and IV products, it is an Anvisa certificate that attests to compliance with Good Practices.
All very well! You can now regularize the colostomy bag
After completing all the previous steps of regularization of the company, you will finally be able to regularize the colostomy bag.
So, in this final phase to obtain Product Registration, it is necessary to present: Labeling, instructions for use, information on possible risks such as: TSS (Toxic Shock Syndrome), maximum use time, information on disposal and others, in addition to the product need to be made of non-toxic material, without fragrance or odor inhibitor.
Even though it is a product that can only be notified, the colostomy bag has extensive and bureaucratic processes, with a series of requirements imposed by Anvisa. If you seek to regularize your company and/or regularize your product, contact Stone Okamont. We have excellent qualified professionals ready to assist and advise you in all this bureaucracy.
Fill out the form below, talk to one of our professionals and get all your questions answered about how to regularize a colostomy bag at Anvisa.
Stone Okamont is also on Youtube. Click here and check out the main subjects related to ANVISA and MAPA.






