Regulatory Services:
ANVISA e MAPA
Advisory and Consulting
ANVISA
View more
Advisory and Consulting
MAPA
View more

Blog
Stone Okamont Stay in!

Plote
Route
Route
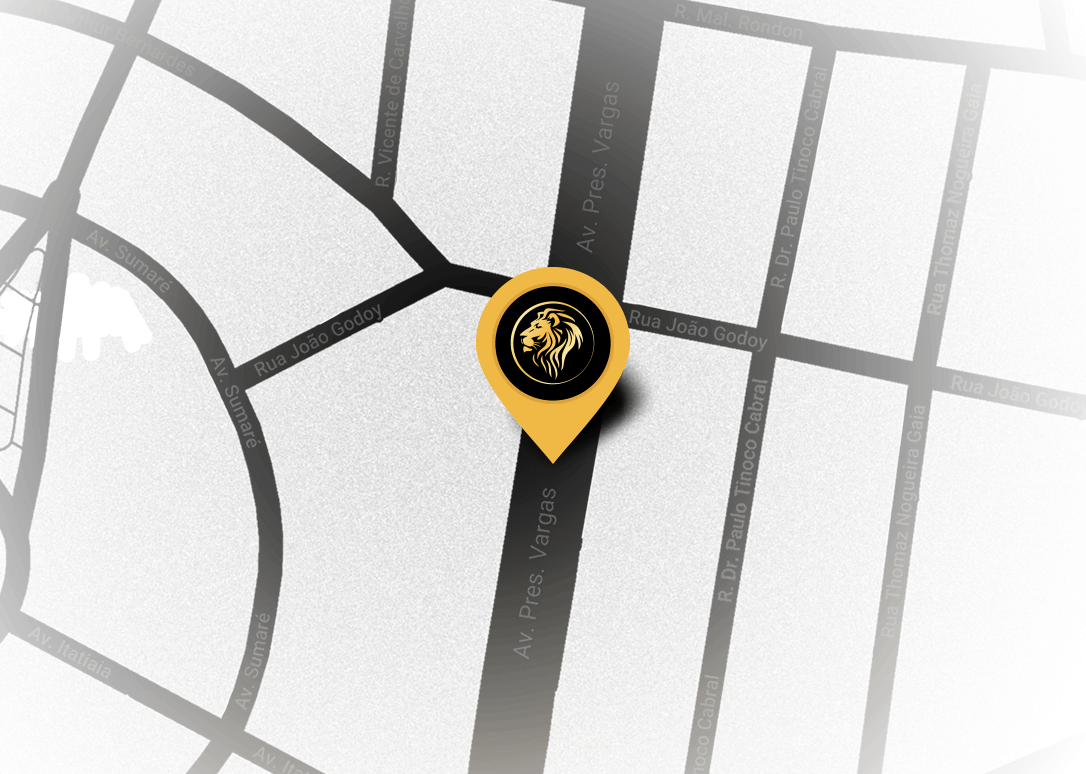
Where
We are
Edifício Times Square
Avenida Presidente Vargas, 2121 - 16° Andar -Sala 1608
Jd. América - Ribeirão Preto/SP - CEP: 14020-260 - Brasil
Jd. América - Ribeirão Preto/SP - CEP: 14020-260 - Brasil
E-mail
Want to know the ideal path
for your company?
Request a quote here.
Be part of
STONE TEAM
If you have knowledge and experience in regulatory affairs, come and be part of the Stone Team!









